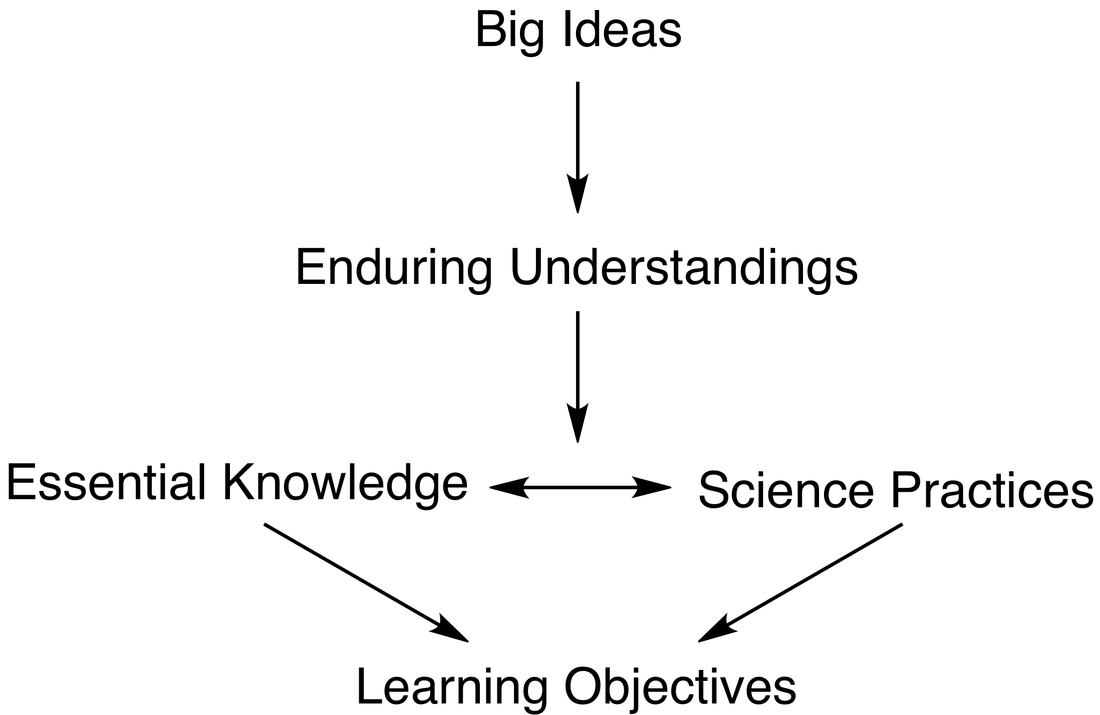
Curriculum Format
The 2014 AP Chemistry curriculum is framed around six Big Ideas:
1. Atoms and Elements - The chemical elements are fundamental building materials of matter, and all matter can
be understood in terms of arrangements of atoms. These atoms retain their identity in chemical reactions.
2. Properties of Matter - Chemical and physical properties of materials can be explained by the structure and the
arrangement of atoms, ions, or molecules and the forces between them.
3. Chemical Reactions - Changes in matter involve the rearrangement and/or reorganization of atoms and/or the
transfer of electrons.
4. Kinetics - Rates of chemical reactions are determined by details of the molecular collisions.
5. Thermodynamic - The laws of thermodynamics describe the essential role of energy and explain and predict the
direction of changes in matter.
6. Equilibrium - Any bond or intermolecular attraction that can be formed can be broken. These two processes are
in a dynamic competition, sensitive to initial conditions and external perturbations.
These Big Ideas are further developed through:
25 Enduring Understanding statements and 71 Essential Knowledge statements about what the student should know.
117 Learning Objectives regarding what the student should be able to do.
7 Science Practices and 25 sub-practices for skills a student should achieve through inquiry.
1. Atoms and Elements - The chemical elements are fundamental building materials of matter, and all matter can
be understood in terms of arrangements of atoms. These atoms retain their identity in chemical reactions.
2. Properties of Matter - Chemical and physical properties of materials can be explained by the structure and the
arrangement of atoms, ions, or molecules and the forces between them.
3. Chemical Reactions - Changes in matter involve the rearrangement and/or reorganization of atoms and/or the
transfer of electrons.
4. Kinetics - Rates of chemical reactions are determined by details of the molecular collisions.
5. Thermodynamic - The laws of thermodynamics describe the essential role of energy and explain and predict the
direction of changes in matter.
6. Equilibrium - Any bond or intermolecular attraction that can be formed can be broken. These two processes are
in a dynamic competition, sensitive to initial conditions and external perturbations.
These Big Ideas are further developed through:
25 Enduring Understanding statements and 71 Essential Knowledge statements about what the student should know.
117 Learning Objectives regarding what the student should be able to do.
7 Science Practices and 25 sub-practices for skills a student should achieve through inquiry.
Example:
|
Big Idea 2: Properties of Matter
Enduring Understanding 2.A: Matter can be described by its physical properties. The physical properties of a substance generally depend on the spacing between the particles (atoms, molecules, ions) that make up the substance and the forces of attraction among them. Essential Knowledge 2.A.3: Solutions are homogenous mixtures in which the physical properties are dependent on the concentration of the solute and the strengths of all interactions among the particles of the solutes and solvent. |
Science Practice 6.2: The student can construct explanations of phenomena based on evidence produced through scientific practices.
Learning objective 2.7: The student is able to explain how solutes can be separated by chromatography based on intermolecular interactions.
Learning objective 2.7: The student is able to explain how solutes can be separated by chromatography based on intermolecular interactions.